Earlier this summer, I introduced you to my research on sulfur in waterlogged wood, and while it hasn’t been all smooth sailing, we’ve made some good headway! After reading up on this problem that plagues many maritime collections and how it’s been tackled in the past, I started conducting experiments to evaluate new approaches for chemical sulfur extraction.
As a quick refresher, when shipwreck timbers sit at the bottom of the ocean, sulfur-reducing bacteria deposit sulfur in the wood. Upon excavation and exposure to oxygen and humidity, accumulated sulfur in the wood can turn into sulfuric acid and sulfur salts. These sulfur compounds degrade the wood and necessitate extensive conservation treatment to achieve stabilization.

To prevent this degradation, scientists and conservators are working to find a way to remove sulfur from waterlogged wood before it dries. We aim to prevent the formation of damaging sulfur products and to decrease the potential for deterioration of the wood.
The Rough Seas of Science
Scientific research helps us learn more about the chemistry of sulfur in waterlogged wood. Solid sulfur is often very stable and unmovable, so we’re trying to find chemicals that can attract sulfur compounds and pull them out of waterlogged wood.

Instead of running chemical tests on actual archaeological wood, we simulate the environment in the lab. I use paper to replicate the chemical structure of waterlogged wood because paper is made from trees. Then, I “contaminate” the paper with the forms of sulfur we know are present in waterlogged wood. Right now, we’re focusing on solid crystals of elemental sulfur and iron sulfide because these particles pose the biggest threat to the wood. (There are other forms of sulfur in waterlogged wood, but they are less threatening and would be more difficult to remove without harming the wood.)
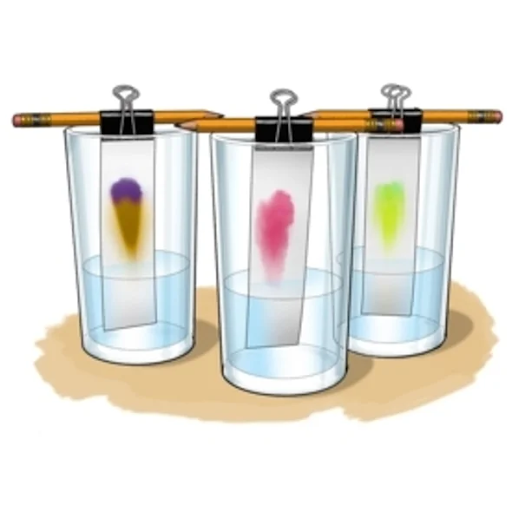
For each experiment, I cut a strip of paper, add sulfur near one end, dip the end in a chemical, and see how sulfur moves through the paper with that chemical. Figuring out this experimental setup was a challenge in itself. Since sulfur is notably stubborn, it was difficult to deposit it on the paper. I also had to figure out how to get the paper to stay upright in the chamber (pictured below). Since the bottom became heavier when it absorbed liquid, the paper tended to fall over. I had to employ some good ol’ tape to secure it, which still fails sometimes, depending on the chemical under inquiry.

Affinity for Water
Despite initial challenges, this simulated system of sulfur on paper now allows us to test sulfur’s attraction to different chemicals through a simple experiment called paper chromatography. Chromatography literally means “color writing,” which you may have experienced if you ever spilled water on paper marked with pen or marker and saw the colors start to spread. The colors separate because the different chemicals that create those colors have different attractions to the water. The colors that travel farther have more attraction, or affinity, for the water, and the colors that travel less far have more relative affinity for the paper.
By using paper chromatography for our experiments, we aim to determine sulfur’s relative affinity for different chemicals versus the paper (aka wood). As sulfur has more affinity for a certain chemical, it will move further up the paper. We can measure how far it travels and compare that distance among different chemicals.
Exploring Our Options
The first chemical I tried was polyethylene glycol (PEG), which is commonly used to stabilize waterlogged wood after it’s dry. PEG is a polymer, which means it’s made of long chains of repeating units. I compared different chain lengths of PEG and different ratios of PEG in water to find the most effective combination.

Next, I tried a few other polymers that have been used in conservation for cleaning organic objects, as well as conservation-appropriate soap. These chemicals have different functional groups, so they interact with sulfur in different ways. I also tried combinations of chemicals to utilize their combined chemical functionalities. Trying new things is what experimentation is all about!
Right now, I’m eager to keep exploring new options and combinations that expand on work others have done. However, there’s always a difference between reading what worked for someone else and trying it yourself! Even with the ideas I’ve gathered from other conservators and scientists, we have to adapt the experiment to our setup and see what actually works in practice.

Conquering Challenges
Inevitably, we encountered more unexpected challenges. How do we see how far the sulfur travels? Paper is white, the chemicals we’re using are mostly colorless, and the amount of sulfur is so small that it’s hard to see with the naked eye. Therefore, I have to dip each paper in a fluorescent dye and view the results under ultraviolet (UV) light. The combination of the dye and UV light makes sulfur stand out against the fluorescent background, so we can see how far it has traveled.
We also used the scanning electron microscope (SEM) at William & Mary’s Applied Research Center to verify sulfur in spots on the paper. The SEM allows super intense magnification to show how sulfur crystals are situated in the paper fibers. This is important for comparisons with previous SEM images of sulfur in wood to ensure our experimental system is accurately representing the sulfur problem in organic objects.

Keeping Hope Afloat
With each new challenge, our experimental process is ultimately improved. Now that I have a reliable experimental protocol, I’m able to efficiently test new chemicals and provide new insights daily. Even chemicals that “don’t work” still offer valuable information about the system. The results of each experiment guide the next step and ensure we keep making progress.
Experimentation is always a bit unpredictable but that’s what makes it worthwhile. The results from these studies will help our team develop a sulfur extraction treatment that can be applied to USS Monitor and other collections of waterlogged wood around the world.